|
The Next Frontier in Audiophile Footer Systems
Printer-Friendly Version (PDF)
Since TuneBlocks were first released in 2005, replacing the standard chromium steel bearing with tungsten carbide has yielded results universally praised by both customers and reviewers. Starting in 2007, Boston Audio Design is offering a new twist on our popular tungsten carbide TuneBlock bearing upgrade – cryogenic tempering. But before discussing this latest innovation, let’s back up and look at how common “footer” systems work, why tungsten carbide is an integral part of many of these systems, and finally – how cryogenic tempering is the next frontier in the evolution of audiophile vibration control (and why we’re patenting the heck out of this process).
How Footer Systems Work
To illustrate how footer systems work in general, let’s take a look at our own TuneBlocks. They work in two ways: first, they minimize the vibration entering the component through contact with the shelf or table it rests upon by absorbing the energy with pure carbon graphite. Graphite (not carbon fiber, with which it is often mistakenly confused) is comprised of horizontal layers of carbon atoms that are loosely bound to each other in the vertical plane, allowing each layer to slide over the other – creating a near infinite number of paths for energy to be converted into heat. Graphite is one of nature’s most amazing materials precisely because its loosely joined molecular structure so readily absorbs energy – be it friction from mechanical parts, heat from nuclear reactors, or minute vibrations coming from transformers or phono cartridges. The unique structure of graphite is also why it has one of the lowest mechanical (also known as “acoustical”) impedances of any material in existence.
Next, and most importantly, TuneBlocks work to remove vibration generated by the component itself. Draining the vibration that comes, for example, from transformers and/or spinning CD mechanisms, while also disrupting and draining chassis resonance, is the key to providing significant performance gains over products that seek only to “isolate” components from the ground.
But exactly how to get the energy from the component and into the graphite block is the main stumbling… well, block, in this process. The solution is to find a material and complementary design that is extremely efficient at conducting energy so that it may be ultimately dissipated by the graphite. The material needs two main properties – hardness and density. The closest “everyday” analogy to this problem is the conductivity of heat. Think about the materials used in your kitchen. When high heat conductivity is needed – for a pot or pan, for example – materials that happen to be very efficient at conducting heat are used such as steel and aluminum. On the other hand, when you want to retain heat, totally different materials are used – such as the ceramic material you’ll find in most coffee cups.
Ironically, these two types of materials are common in the hifi “isolation” industry because each forms half of the necessary hardness/density equation: steel is relatively dense, but is not that hard on an absolute scale (not to say that it won’t hurt if a big bar of it hits you on the head!), and ceramic is extremely hard, but not very dense.
The material that bridges this divide is tungsten carbide, which is nearly as hard as the hardest ceramics and is much denser than steel. Boston Audio Design, along with other leading audiophile accessory manufacturers, has used tungsten carbide for years. But before describing our latest innovation in the cryogenic tempering of tungsten carbide, let’s first look at the other ways that tungsten carbide has been used by other companies.
Popular Approaches of Bearing Footer Systems
When you get beyond entry-level footer systems (made from sorbothane, rubber, cork, etc.) that merely try to “isolate” components without providing an interface to absorb component-generated resonance, many manufacturers use a similar vibration control principle – that the hardness/density of a particular bearing material, combined with the freedom of movement that comes from placing the bearing in a “race” or polished cup, is sufficient for dissipating vibration. Boston Audio Design completely rejects this theory. It is true that this design is effective at reducing some types of vibration and has been used in architectural and industrial applications for decades. But the notion that a ball can vibrate and roll at the same frequencies as are present in audio equipment and thus “dissipate the energy as heat” seems, at best, a partial solution.
The entire premise of the theory of operation of such systems is that the finely polished bearing can be in contact with another similarly polished surface that, together, permits energy dissipation as heat through lateral motion. We would concede that, in theory, this could work as proposed (provided that the base cup was absolutely vibration-free, otherwise shelf vibration would diminish efficacy) – but only in a perfect vacuum. For those of us living within the Earth’s atmosphere, it is simply inevitable that dust, dirt and corrosion invade the ball/cup interface and throw a wrench into those finely polished works. To say that roundness alone will effectively dissipate energy though lateral motion is just unrealistic since the surfaces themselves are “corrupted” the instant we put our greasy human fingers on them… and more so as dirt, dust and corrosion inevitably encapsulate them over time. The bearings may be “Grade 3”, but what about the stuff on them?
Boston Audio Design’s Approach to the Bearing Interface
Boston Audio Design’s approach to tungsten carbide is to use the bearings as a stationary coupler between the equipment and the graphite material. As opposed to other designs that employ bearings, TuneBlocks are unique in that they do not “wobble” or in any way imperil the safety of your components. Rather than attempting to dissipate energy through the movement of a bearing in a cup, we simply found a better material – a material that itself dissipates vibration on a molecular level. As we discussed earlier, graphite is made of nearly infinite 1-atom thick layers of carbon, each of which is capable of sliding back and forth over adjacent layers, thus effectively dissipating energy without leaving a sonic signature of its own (which – in our opinion – is the result of graphite’s incredibly low mechanical impedance, as mentioned above).
So why use bearings at all? Why not just use a chunk of tungsten carbide? There are a couple of reasons why we have chosen to use tungsten carbide bearings. First, while tungsten carbide bearings are very expensive to manufacture, they are considerably less expensive than manufacturing custom shapes – shapes that would achieve the same purpose. This helps us keep retail costs as reasonable as possible for the customer. Second, a smoothly polished finish is an important (but not the only important) performance parameter. Our highly polished tungsten carbide bearing is more than sufficient for maintaining outstanding surface contact with the component being treated and with the graphite block. Again, to produce an alternate shape other than a sphere at these kinds of tolerances would be prohibitively expensive for no performance gain.
The quest to take our TuneBlocks to the next level has led us to search in some pretty interesting directions – including a variety of very expensive tungsten carbide alloys that, while offering good results, ultimately proved a dead end because of cost ($400 bearing upgrade anyone?). Knowing that cryogenic tempering has been used for decades with ferrous metals to improve hardness and wear resistance in industrial applications, we started researching the cryogenic tempering of tungsten carbide. Since tungsten carbide does not contain iron – the key component of the ferrous metals that have traditionally benefited from cryogenic tempering – we were curious as to whether it was even worth trying at all. But what exactly is cryogenic tempering?
Understanding Cryogenic Tempering
Frankly, the first thing that I (and probably a lot of other baseball fans) thought of when I heard the word “cryogenic” is the Boston Redsox legend Ted Williams, whose dismembered head was embroiled in a legal dispute between family members as to whether it should be cryogenically frozen in hopes of future revival. It turns out that the fringe practice known as “cryonics” is totally different from the proven industrial applications of deep freezing metals… and is more than a little wacky in my opinion!
The science of cryogenics dates back to WWII, when researchers found that super cooling steel increased both hardness and wear resistance. These researchers simply dipped steel into liquid nitrogen (-320 degrees Fahrenheit) and noticed that while wear resistance was improved, the treated materials were more fragile and prone to breakage (something now called “thermal shock”). Fast forward to today and cryogenic processing, through further testing and refinement, has become a widely used, predictable way to increase wear resistance with few (if any) of the earlier problems. In fact, cryogenic treatment has now been available commercially for over 40 years. Cryogenic processing is widely used for a host of applications including scientific research, liquefying natural gas, creating special fuel for rockets, and improving the performance of machine tools.
Just when does “cold” become “cryogenic?” The difference between simple refrigeration and cryogenic processing is simply a matter of degree (excuse the pun): true cryogenic freezing occurs around 300 degrees below zero Fahrenheit (-180 degrees Celsius) – well below the limits of common refrigerants such as Freon. Most cryogenic processing today is preformed with liquid nitrogen in a computer controlled environment for precise cooling and restoration to ambient temperature.
For industrial applications – such as improving the performance of drill bits and end mills – the benefits of cryogenic processing are indisputable, just as are the benefits of heat-treating (such as quenching, in which steel is heated and then rapidly cooled). Depending on the exact type of steel being treated, tool life can be extended two to five times or even more. Exactly how these results are achieved can still be a source of controversy (especially in non-ferrous metals), but the most widely accepted explanation is that, in ferrous metals, the extreme cold changes the way the carbon atoms relate to the ferrous atoms in the metal’s crystal lattice structure. Namely, the metal’s retained austenite is changed to martenstite, yielding a denser crystal lattice structure.
To get the meaning of the italicized sentence above, let’s think first back to chemistry class. A metal is an element that freely shares electrons among a three-dimensional “lattice” of metal atoms. Because metal atoms are able to share electrons, they are able to conduct heat and electricity. For example, copper is a well-known element that excels at conducting heat (copper posts and pans are revered among chefs) and electricity (the ubiquitous copper wire conducts most of the electricity that makes modern life possible).
Steel is a metal alloy since it combines a number of different elements, but the principle is the same as our copper example – atoms arranged in a lattice structure that freely share electrons. Steel is made of a combination of carbon and iron (the iron makes it a “ferrous” metal). Different alloys of steel change the proportion of those elements or even add additional elements to the mix. For us the point is to understand how the atoms are bound together.
The lattice structure is simply the 3-dimensional atomic shape of the material. All those photons, neutrons and electrons have to go somewhere, and the lattice structure assigns each a place in the correct structure in space. One of the most telling examples of how lattice structure affects material properties is to consider the difference between diamond and graphite (one of the hardest natural materials and one of the softest, respectively). Both are made of the exact same carbon atoms, but the difference is in how those atoms are held together. Diamonds have a very strong lattice structure arranging each atom, making it very difficult for a group of atoms to let one of its members escape. On the other hand, graphite is made of a layered lattice structure. Graphite’s atoms are bound together strongly on one plane, but weakly on another – making the layers prone to sliding on top of each other. Think of a pencil lead (actually a form of graphite) – just rubbing it lightly on just about anything leaves a string of abandoned carbon atoms.
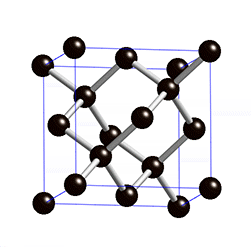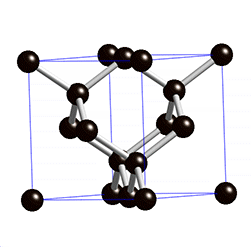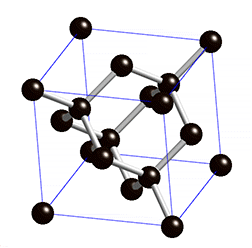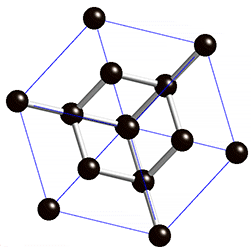
Diamond lattice. | 
Graphite lattice. |
To revisit our sentence describing how cryogenic tempering works in steel: the metal’s retained austenite is changed to martenstite, yielding a denser crystal lattice structure.
Now that we understand what a “crystal lattice structure” is, let’s understand the rest. Unlike diamond and graphite, which are made from the single element carbon, steel is an alloy made of carbon and iron. The way that (some of) these atoms are arranged in a lattice is what is meant by “austenite” and “martenstite.” Austenite is part of the structure of most steels and is a simple square shape. In fact, austenite is more of a byproduct of steel production, since most of it is eliminated during heat treatment as the iron and carbon are transformed into a martenstite structure, which is not only larger and stronger, but is rectangular in shape.
Here’s where the cryogenic tempering enters the picture – by super-cooling the material, the “retained austenite is changed to martenstite, yielding a denser crystal lattice structure.”
Ideally, you now understand how cryogenic tempering works! Congratulations. What remains after treatment in the case of steel is a much stronger, denser atomic structure – sort of completing the transformation that simple heat-treating is unable to do. Further, by removing retained austenite, the crystal structure is more consistent and retained stress between the austenite and martenstite is removed, leaving behind a number of mechanical benefits in addition to increased hardness.
Cryogenically Tempering Tungsten Carbide
Now back to tungsten carbide and how cryogenic tempering can have tremendously positive effects for audiophile applications. With your possibly newfound understanding of cryogenically treating steel, you may ask – “ok, but if cryogenic tempering works by transforming the relationship between iron and carbon, where’s the iron in tungsten carbide?” That is a great question!
In fact, there is no iron in tungsten carbide, but there is something in addition to carbon and tungsten – cobalt. In order to render pure tungsten carbide less brittle, “tungsten carbide” materials such as the bearings used in TuneBlocks are actually “cemented carbides,” or tungsten carbide particles suspended within a cobalt matrix. Recent studies at Pennsylvania State University and Arizona State University demonstrate conclusively that cryogenically tempering tungsten carbide improves durability on an order of magnitude similar to increases in steel strength – pretty incredible considering that tungsten carbide is already incredibly durable.
Exactly why cryogenically treating tungsten carbide works is still up for debate. One theory is that since cobalt is next to iron on the Periodic Table and has similar properties at the atomic level (including lattice structures), it possibly reacts in the same way that iron reacts to changes in temperature. Other researchers point to a reduction in residual stress in the material as the potential cause of the increased wear resistance.
When we at Boston Audio Design started researching the cryogenic tempering of tungsten carbide, we contacted one of the most respected cryogenic labs in the country and were lucky to find someone at the company who was also interested in the audiophile applications of cryogenic treatment. In fact, he tells us that some manufacturers send their vacuum tubes to his company for treatment (a trade secret of at least a few equipment manufacturers apparently). While it may seem from this article that “cryogenic tempering” is just one process, there are in fact many variations of the technique that include not only the destination temperature, but how long the material is held at the temperature, how quickly it is cooled, how quickly it is warmed, and whether additional heat is applied after cooling.
We tried a variety of cryogenic techniques and determined that one in particular gave the results we were looking for. The result was nothing less than the first commercially viable performance improvement we have been able to find to the component/footer interface. We’re still exploring exactly why our cryogenic process yields the results that it does, but we suspect that the same mechanisms that make tungsten carbide stronger as the result of treatment (changes to the cobalt binder, changes in the crystal lattice structure) also affect the material’s ability to conduct vibration from the components and into the graphite block. As we learned earlier in this paper, the more efficient the medium for channeling energy, the more effective graphite can be at dissipating it.
By adapting some of the most recent advances in cryogenic tempering and applying them to audiophile ends, we are able to bring to market our highest performance TuneBlock yet – the SE. With more than twice the graphite mass of the standard TuneBlock and a 50% larger bearing that has been cryogenically tempered according to our emersion process, we feel that the SE represents the logical fulfillment of the TuneBlock design… and one of the highest performance footer systems available.
Boston Audio Design was the first to bring pure carbon graphite and tungsten carbide together into a high performance audio product. We will continue to search for the “next frontier” in audiophile performance – wherever we find it.
|